Paint coatings
Paint is the most commonly used material to protect steel. Paint systems for steel structures have developed over the years to comply with industrial environmental legislation and in response to demands from bridge and building owners for improved durability performance. Previous five and six coat systems have been replaced by typically three coat alternatives, and the latest formulations have focussed on application in even fewer numbers of coats, but with increasing individual film thickness. Examples of this are epoxy and polyester glass flake coatings that are designed for high build thickness in one or two coat applications, and single coat high build elastomeric urethane coatings, up to 1000μm thick.
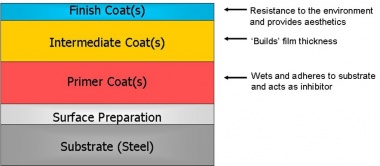
Modern specifications usually comprise a sequential coating application of paints or alternatively paints applied over metal coatings to form a ‘duplex’ coating system. The protective paint systems usually consist of primer, undercoat(s) and finish coats. Each coating ‘layer’ in any protective system has a specific function, and the different types are applied in a particular sequence of primer followed by intermediate / build coats in the shop, and finally the finish or top coat either in the shop or on site.
[top]Composition of paints and film formation
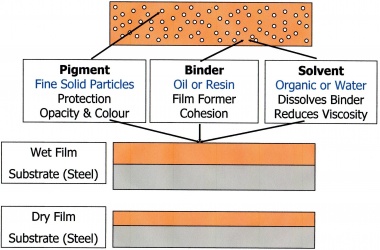
Paints are made by mixing and blending three main components:
(a) The pigments
Pigments are finely ground inorganic or organic powders which provide colour, opacity, film cohesion and sometimes corrosion inhibition.
(b) The binder
Binders are usually resins or oils but can be inorganic compounds such as soluble silicates. The binder is the film forming component in the paint.
(c) The solvent
Solvents are used to dissolve the binder and to facilitate application at the paint. Solvents are usually organic liquids or water.
Paints are applied to steel surfaces by many methods but in all cases this produces a 'wet film'. The thickness of the 'wet film' can be measured, before the solvent evaporates, using a comb-gauge. As the solvent evaporates, film formation occurs, leaving the binder and pigments on the surface as a 'dry film'. The thickness of the 'dry film' can be measured, usually with an electro-magnetic induction gauge. The relationship between the applied 'wet film' thickness and the final 'dry film' thicknesses (dft) is determined by the percentage volume solids of the paint, i.e. dft = 'wet film' thickness multiplied by the % vol. solids.
In general the corrosion protection afforded by a paint film is directly proportional to its dry film thickness.
[top]Classification of paints
Since, in the broadest terms, a paint consists of a particular pigment, dispersed in a particular binder, dissolved in a particular solvent then the number of generic types of paint is limited. The most common methods of classifying paints are either by their pigmentation or by their binder type.
Primers for steel are usually classified according to the main corrosion inhibitive pigments used in their formulation, e.g. zinc phosphate primers and metallic zinc primers, etc. Each of these inhibitive pigments can be incorporated into a range of binder resins giving for example, zinc phosphate alkyd primers, zinc phosphate epoxy primers, etc.
Intermediate and finish coats are usually classified according to their binders, e.g. epoxies, vinyls, urethanes, etc.
[top]Paint coatings
Paints are usually applied one coat on top of another and each coat has a specific function / purpose. These are described as follows.
[top]Primers
The primer is applied directly onto the cleaned steel surface or, in the case of duplex systems, the sealed metal coating. Its purpose is to wet the surface and to provide good adhesion for subsequently applied coats. For primers applied directly to steel surfaces, these are also usually required to provide corrosion inhibition. There are two basic types of primer.
(a) Primers pigmented with metallic elements anodic to steel
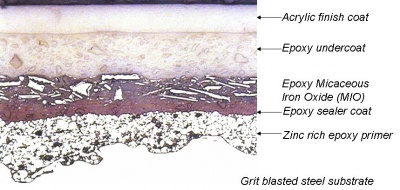
These primers are formulated so that, when a break in the coating (due to damage or local corrosion) exposes the steel substrate, the anodic metal corrodes sacrificially in preference to the steel. This effectively stifles steel corrosion and under-rusting of the primer until the anodic metal is exhausted. Zinc-rich primers are the most commonly used of this type.
(b) Primers relying on the high adhesion and chemical-resistance properties of the binder
With these primers, good adhesion is obtained (provided that the surface is very thoroughly cleaned) and it is sufficient to prevent under-rusting at any break in the coating (due to damage). Two-pack epoxy primers are typical of this type. These primers may contain inhibitive pigments to interfere with the corrosion process. Zinc phosphate, for example, is a mildly inhibitive pigment and is widely used in modern primer formulations.
[top]Intermediate coats
Intermediate coats are applied to ‘build’ the total film thickness of the system. Generally, the thicker the coating the longer the life. Intermediate coats are specially designed to enhance the overall protection and, when highly pigmented, decrease permeability to oxygen and water. The incorporation of laminar pigments, such as micaceous iron oxide (MIO), reduces or delays moisture penetration in humid atmospheres and improves tensile strength. Modern specifications now include inert pigments such as glass flakes to act as laminar pigments. Undercoats must remain compatible with finishing coats when there are unavoidable delays in applying them.
[top]Finish coat
The finish coat provides the required appearance and surface resistance of the system. Depending on the conditions of exposure, it must also provide the first line of defence against weather and sunlight, open exposure, and condensation.
[top]Stripe coats
Stripe coats are additional coats of paint that are applied locally to welds, fasteners and external corners. Their function is to build a satisfactory coating thickness at edges and corners where paint has a tendency to contract and thin upon drying. Specifications should indicate the type and number of stripe coats required and state when they are to be applied.
[top]The paint system
The various superimposed coats within a painting system have, of course, to be compatible with one another. They may be all of the same generic type or may be different, e.g. chemical resistant types, such as a recoatable polyurethane finish coat, may be applied onto epoxy primer and intermediate coats. However, as a first precaution, all paints within a system should normally be obtained from the same manufacturer and used in accordance with the manufacturer’s recommendations.
An important factor in the coating system is the definition and measurement of the dry film thickness (dft). Dry film thicknesses are generally checked on the complete paint system, although individual films may be checked separately. Usually, nominal dry film thicknesses are specified but sometimes minimum values are quoted.
For nominal dry film thicknesses, individual values less than 80% of the nominal thickness are not acceptable. Values between 80% and 100% are acceptable provided that the overall average (mean value) is equal to or greater than the nominal.
Specifications for minimum dry film thicknesses require careful paint application to avoid excessive film thickness. The ‘over application’ of paints can result in the formation of high stresses and may cause premature failure of the system. Wet film thickness (wft) checks may also be required during the application of the coating to check that a subsequent satisfactory dry film thickness will be achieved.
[top]Main generic types of paint and their properties
(a) Air drying paints
For example alkyds
These materials dry and form a film by an oxidative process, which involves absorption of oxygen from the atmosphere. They are therefore limited to relatively thin films. Once the film has formed it has limited solvent resistance and usually poor chemical resistance.
(b) One pack chemical resistant paints
For example acrylated rubbers, vinyls
For these materials, film formation requires only solvent evaporation and no oxidative process is involved. They can be applied as moderately thick films though retention of solvent in the film can be a problem at the upper end of this range. The formed film remains relatively soft and has poor solvent resistance but good chemical resistance. Bituminous paints also dry by solvent evaporation. They are essentially solutions of either asphaltic bitumen or coal-tar pitch in organic solvents.
(c) Two pack chemical resistant paints
For example epoxy, urethane
These materials are supplied as two separate components, usually referred to as the base and the curing agent. When these two components are mixed, immediately before use, a chemical reaction occurs. These materials therefore have a limited 'pot life' before which the mixed coating must be applied. The polymerisation reaction continues after the paint has been applied and after the solvent has evaporated to produce a densely cross linked film which can be very hard and has good solvent and chemical resistance. Liquid resins of low viscosity can be used in the formulation thereby avoiding the need for a solvent. Such coatings are referred to as 'solvent less' or 'solvent free' and can be applied as very thick films.
| Binder | System cost | Tolerance of poor surface | Chemical resistance | Solvent resistance | Water resistance | Overcoating after aging | Comments |
|---|---|---|---|---|---|---|---|
| Black Coatings (based on Tar products) | Low | Good | Moderate | Poor | Good | Very good with coatings of same type | Limited to black or dark colours. May soften in hot conditions. |
| Alkyds | Low – Medium | Moderate | Poor | Poor – Moderate | Moderate | Good | Good decorative properties. High solvent levels. |
| Acrylated Rubbers | Medium – High | Poor | Good | Poor | Good | Good | High build films that remain soft and are susceptible to sticking. |
| Epoxy (Surface Tolerant) |
Medium – High | Good | Good | Good | Good | Good | Can be applied to a range of surfaces and coatings. |
| Epoxy (High performance) |
Medium – High | Very Poor | Very Good | Good | Very Good | Poor | Susceptible to ‘chalking’ in U.V. light. |
| Urethane and Polyurethane | High | Very Poor | Very Good | Good | Very Good | Poor | Can be more decorative than epoxies. |
| Organic Silicate and Inorganic Silicate | High | Very Poor | Moderate | Good | Good | Moderate | May require special surface preparation |
[top]Prefabrication primers
Also referred to as blast primers, shop primers, temporary primers, holding primers, etc., these primers are sometimes used on structural steelwork, immediately after blast cleaning, to maintain the reactive blast cleaned surface in a rust free condition until final painting can be undertaken. They are mainly applied to steel plates and sections before fabrication. The main requirements of a prefabrication primer are as follows:
- The primer should be capable of airless spray application to produce a very thin even coating. Dry film thickness is usually limited between 15-25μm. Below 15μm, the peaks of the blast profile are not protected and 'rust rashing' occurs on weathering. Above 25μm, the primer affects the quality of the weld and produces excessive weld fume.
- The primer must dry very quickly. Priming is often done in-line with automatic blast cleaning plant which may be handling plates and/or sections at a pass rate of between 1-3 metres/minute. The interval between priming and handling is usually of the order of 1-10 minutes and hence the primer film must dry within this time.
- Normal fabrication procedures (e.g. welding, gas cutting) must not be significantly impeded by the coating, and the primer should not cause excessive weld porosity. A welding certificate should be available from the paint manufacturer.
- Weld fume omitted by the primer must not exceed the appropriate Occupational Exposure Limits. Proprietary primers are tested and certified by the Newcastle Occupational Health Agency. A health and safety certificate should be available from the paint manufacturer.
- The primer coating should provide adequate protection until either overcoated or placed within a non corrosive environment e.g. indoor storage or within a building construction. In practice, such periods are rarely met except in the least arduous conditions, e.g. indoor storage. In aggressive conditions, durability can often be measured in weeks rather than months. Zinc rich and zinc silicate primers provide the highest order of protection of all prefabrication primers.
- The primed surface, after weathering, should require the minimum of re-preparation for subsequent painting and must be compatible with the intended paint system. Many proprietary prefabrication primers are available but they can be classified under the following main generic types:
[top]Etch primers
These are based on polyvinyl butyral resin reinforced with a phenolic resin to increase water resistance. These primers can be supplied in a single pack or two pack form, the latter providing better durability.
[top]Epoxy primers
These are two pack materials utilising epoxy resins and usually have either polyamide or polyamine curing agents. They are pigmented with a variety of inhibitive and non-inhibitive pigments. Zinc phosphate epoxy primers are the most frequently encountered and give the best durability within the group.
[top]Zinc epoxy primers
These primers can be either zinc rich or reduced zinc types. Zinc rich primers produce films which contain about 85% by weight of metallic zinc powder and the reduced zinc type as low as 55% by weight. When exposed in either marine or highly industrial environments, zinc epoxy primers are prone to the formation of insoluble white zinc corrosion products which must be removed from the surface before subsequent overcoating.
[top]Zinc silicate primers
Zinc silicate primers produce a level of protection which is comparable with the zinc rich epoxy types and they suffer from the same drawbacks, e.g. formation of zinc salts and production of zinc oxide fume during welding. There are currently different categories of zinc silicate primers based upon the binder (organic or inorganic) and the zinc content. Low zinc primers in this group have been developed to improve their weldability and to minimise weld porosity, however their durability is also reduced. The organic silicate primers are the most suitable as prefabrication primers.
[top]Application of paint coatings
[top]Methods
The method of application and the conditions under which paints are applied have a significant effect on the quality and durability of the coating. Standard methods used to apply paints to structural steelwork include application by brush, roller, conventional air spray and airless spray.
Airless spraying has become the most commonly used method of applying paint coatings to structural steelwork under controlled shop conditions. Brush and roller application are more commonly used for site application, though spraying methods are also used.
[top]Brushing
This is the simplest method and also the slowest and therefore most expensive. Nevertheless it has certain advantages over the other methods, e.g. better wetting of the surface and can be used in restricted spaces, be useful for small areas, with less wastage and contamination of surroundings.
Brush application of paint
(Video courtesy of Corrodere/MPI)
[top]Roller
This process is much quicker than brushing and is used for large flat areas, but demands suitable rheological properties of the paint.
Roller application of paint
(Video courtesy of Corrodere/MPI)
[top]Air spray
Paint coatings for structural steelwork are usually spray applied. The paint is atomised into fine droplets and projected onto the surface to be protected where the droplets join together to form a continuous film. The atomisation can be accomplished in a number of ways.
In air spraying, the paint is atomised by mixing it with a stream of compressed air in a conventional spray gun. The paint can be either sucked into the air stream (as in the simple suction-cup gun used for application to small areas) or fed to the spray gun under pressure from a pressure pot. For ideal application, careful adjustments of the spray nozzle and air pressures must be made by a skilled operator, according to the consistency and composition of the paint product and the film thickness required. The application rates for air spray are quicker than for brushing, or rolling, however paint wastage by overspray is high.
[top]Airless spray
For airless spraying, the paint is hydraulically compressed and, on release through a small orifice in an airless spray gun, it is atomised and projected onto the surface. By changing the orifice size and shape and by varying the hydraulic pressure, atomisation can be accomplished for a wide range of paint consistencies from thin to thick, to give a wide range of rates of deposition. The equipment required is much more expensive than for conventional air spraying, because it must withstand the much higher pressures involved. However, the application rates are higher than for air spray with overspray wastage greatly reduced. For conventional air spraying, the maximum air pressure will normally not exceed 100 psi (6.9 bar); for airless spraying, hydraulic pressures up to 4,000 psi (280 bar) may be required.
A variant of the above involves heating to reduce the consistency of the paint rather than adding diluents. In this way greater film thickness per application is achieved. This method can be used for the application of solvent-free materials such as two-pack products, which can be mixed at the spray gun nozzle at the moment of application. The use of expensive equipment and highly skilled labour is necessary for the achievement of optimum results but may be justified for the protection of large and important structures.
Airless paint spraying
(Video courtesy of Corrodere/MPI)
[top]Conditions of application
The principal conditions that affect the application of paint coatings are temperature and humidity. These can be more easily controlled under shop conditions than on site.
(a) Temperature
Air temperature and steel temperature affect solvent evaporation, brushing and spraying properties, drying and curing times and the pot life of two-pack materials, etc. Where heating is required, this should only be by indirect methods.
(b) Humidity
Paints should not be applied when there is condensation present on the steel surface or the relative humidity of the atmosphere is such that it will affect the application or drying of the coating. Normal practice is to measure the steel temperature with a contact thermometer and to ensure that it is maintained at least 3°C above the dew point.
However, moisture cured paints are available. These paints are specifically formulated for application in damp and humid conditions; reference should be made to the manufacturer’s data sheets for details of limiting conditions of application.
[top]Coating applicator training and certification (ICATS)
With modern high performance coatings, correct application has become increasingly important to achieve the intended performance. Industry has recognised this and established a training and certification scheme for paint applicators (ICATS – Industrial Coating Applicator Training Scheme). ICATS registration (or to an equivalent scheme) has subsequently become a mandatory requirement for work on National Highways and Network Rail bridges.
ICATS is owned by the Institute of Corrosion and operated through Correx Ltd., a wholly owned subsidiary of the Institute. The scheme has a mandatory Basic Unit 'Industrial Coating Applicator' that comprises 6 modules:
- Health & Safety
- Site Access
- Plant & Equipment
- Surface preparation
- Paint Types & Application
- Quality Control
There are also 2 optional specialist modules:
- Abrasive Blast Cleaning
- Spray Painting
ICATS is a registered training scheme that meets the requirements of NHSS 19A[1] and enables contractors to provide 'best value' service to maximise coating performance over structure life and to reduce whole life costs.
[top]Additional education
For engineers and inspectors wishing to further their knowledge of corrosion control in the context of structural steelwork, there are web based training programmes available from a specialist training provider, Corrodere:
- Trainthepainter
- Traintheinspector
- Trainthecoatingspecialist
Trainthepainter is an internationally recognised and certified training programme that provides trainee coating applicators with a comprehensive package of training modules that ensure professionalism and give an insight into methods of surface treatment and application of protective coatings. As with ICATS, this is a registered training scheme that meets the requirements of NHSS 19A[1].
[top]Relevant Steel for Life sponsors
This website is maintained by Steel for Life with funding provided by a number of BCSA’s Industry Members. Those sponsors relevant to this article are as follows:
Silver
[top]References
[top]Resources
- Hendy, C.R.; Iles, D.C. (2015) Steel Bridge Group: Guidance Notes on best practice in steel bridge construction (6th Issue). (P185). SCI
- Steel Buildings, 2003, The British Constructional Steelwork Association Ltd.
- Chapter 12 – Corrosion Protection
[top]Further reading
- D.Deacon & R.Hudson (2012), Steel Designer’s Manual (7th Edition), Chapter 36 - Corrosion and corrosion prevention, The Steel Construction Institute.
- D.A. Bayliss & D.H.Deacon (2002), Steelwork Corrosion Control (2nd edition), Spon Press.
[top]See also
- Corrosion of structural steel
- Influence of design on corrosion
- Surface preparation
- Standard corrosion protection systems for buildings
- Metallic coatings
- Appropriate specifications
- Inspection and quality control